Xinyan Medical at CMEF Spring 2026 – Visit Us at Booth 6.1D07!
April 9–12, 2026 | National Exhibition and Convention Center, Shanghai | Booth: 6.1D07
Dear Global Medical Industry Colleagues and Partners,
Xinyan Medical cordially invites you to CMEF Spring 2026 (the 93rd China International Medical Equipment Fair). As a high-tech enterprise specializing in intelligent ECG monitoring, we will showcase our full-series wearable ECG devices, multi-parameter integrated system solutions, and veterinary-grade monitoring equipment at Booth 6.1D07.
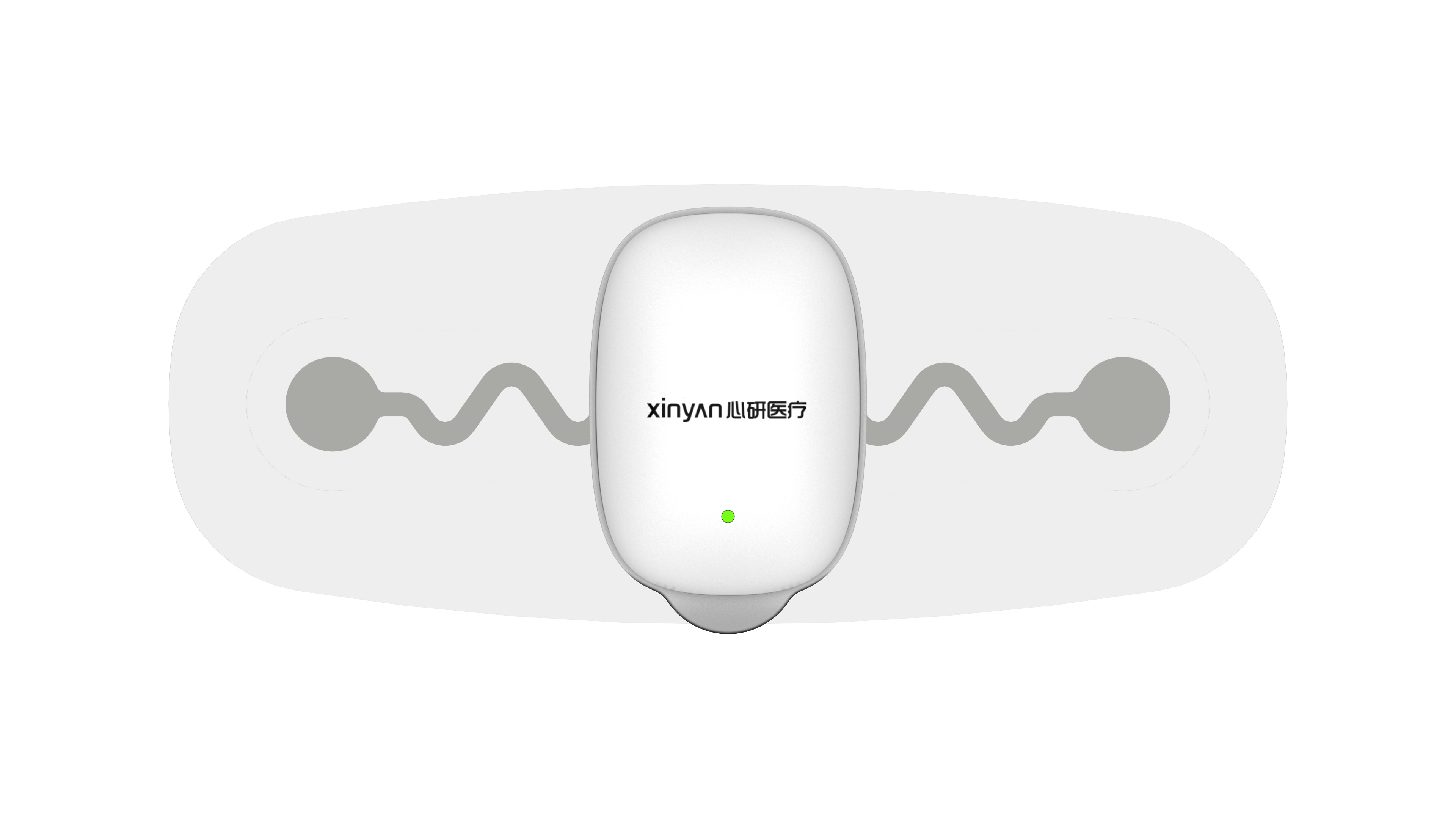
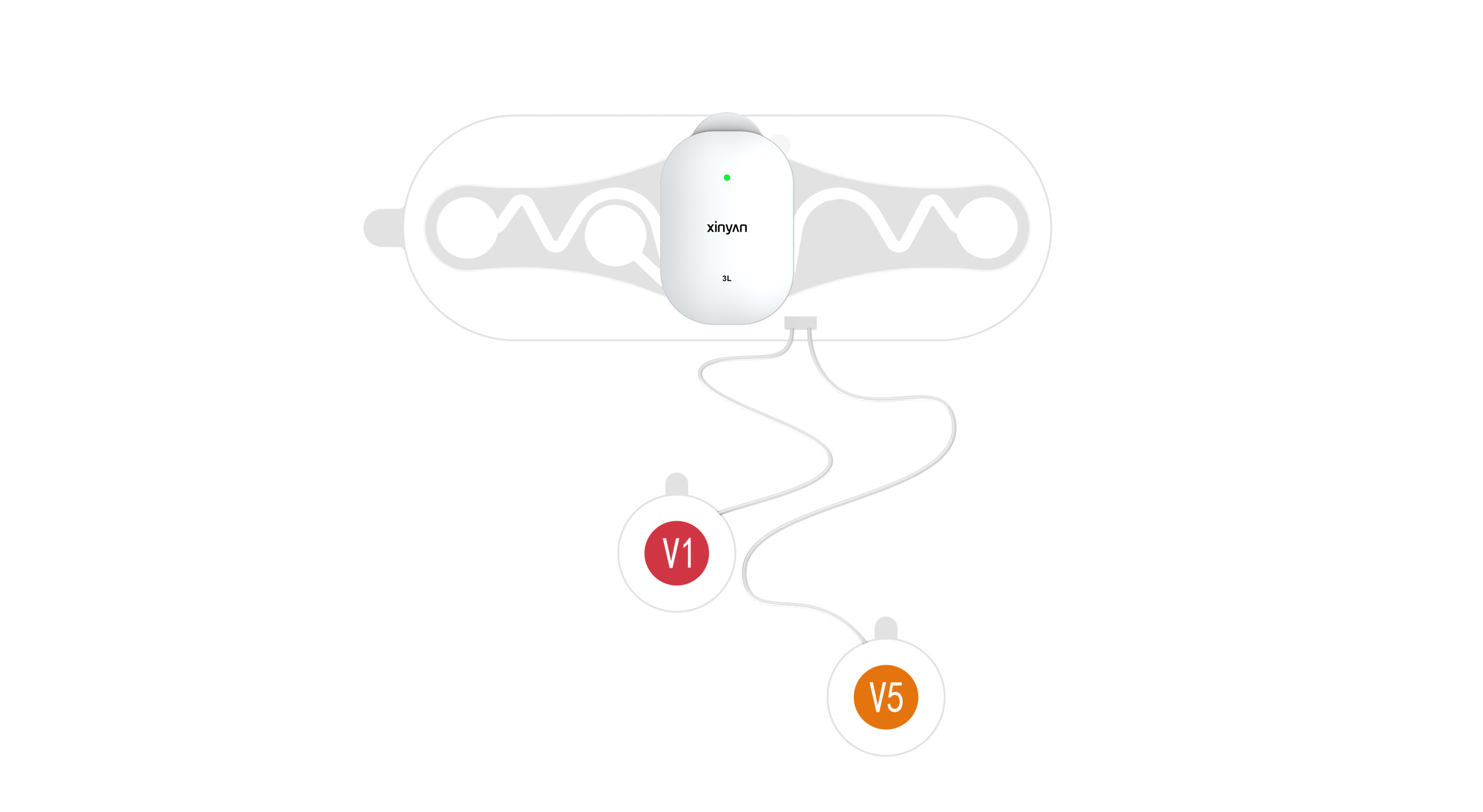
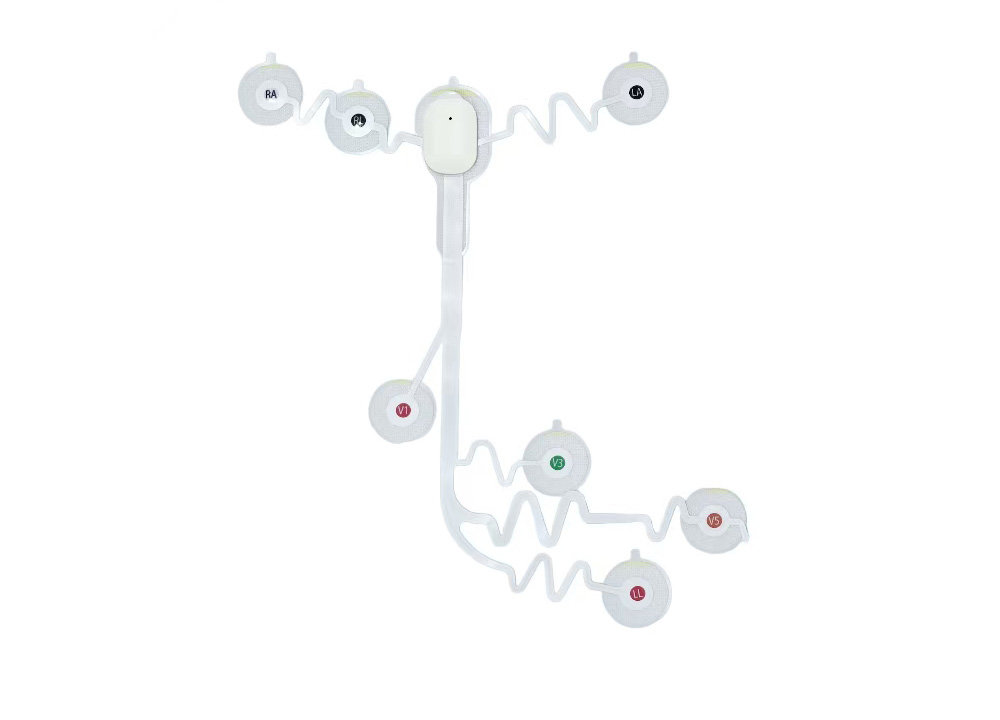
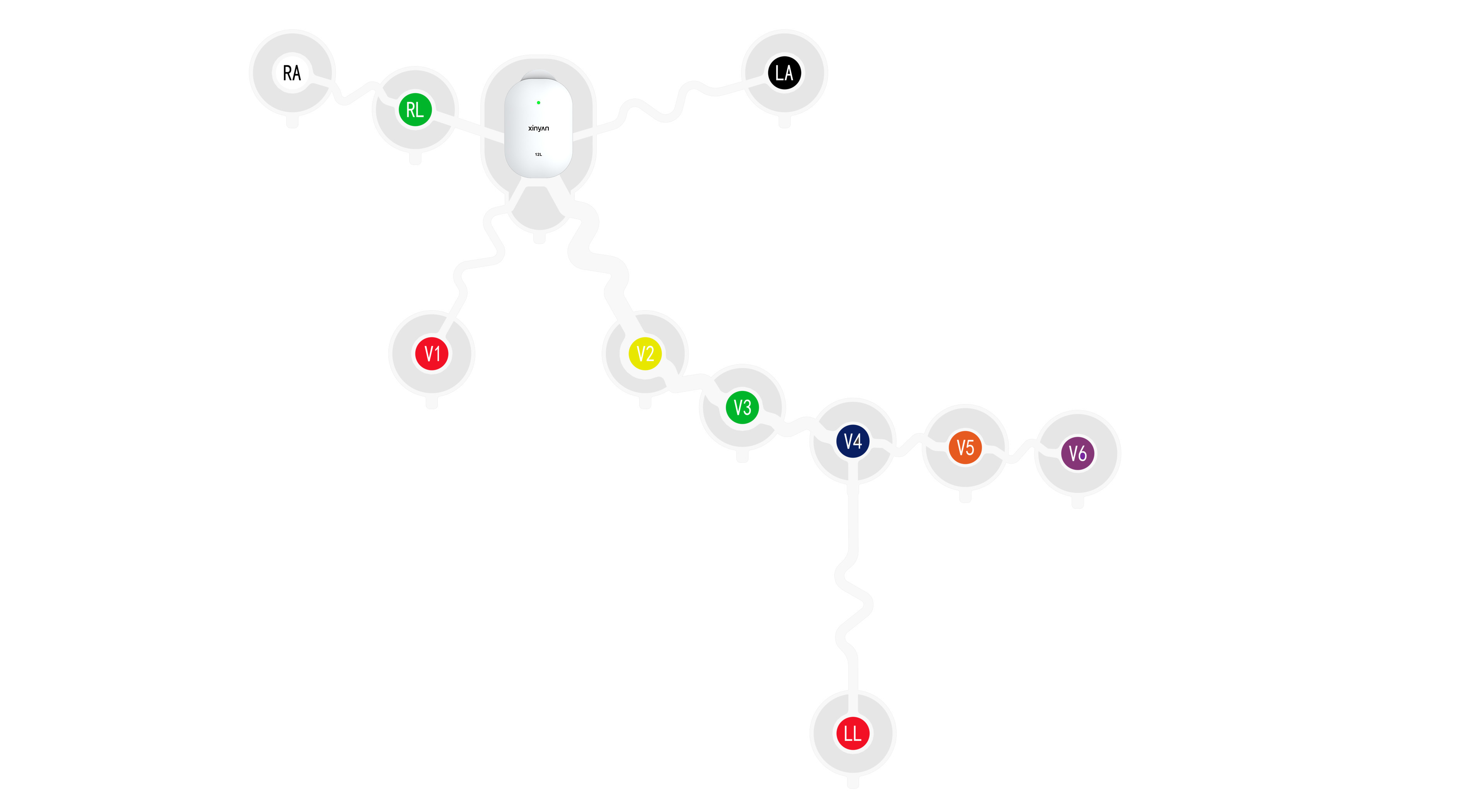
1. Wearable ECG for Human Use
Full coverage from single-lead to 12-lead devices, supporting 7‑day long‑term monitoring, IP27 dust/water resistance, and battery‑free design. Suitable for home screening, community healthcare, and clinical diagnosis.
2. Multi-Parameter System Solutions
Based on our ECG technology platform, we demonstrate integrated parameters including body temperature, respiration, posture, SpO₂, and CGM. Features a "cockpit‑style" data platform for real‑time patient overview, applicable for in‑hospital monitoring, 6‑minute walk tests, rehabilitation management, and chronic disease care.
3. Veterinary Multi-Parameter Monitoring
YMHeart Vet series, designed for dogs and cats, supports simultaneous monitoring of ECG, temperature, respiration, and posture. Medical‑grade precision for pre‑operative assessment, intra‑operative monitoring, post‑operative evaluation, and geriatric pet care.
4. AI-Powered Platform
Remote monitoring system with AI‑assisted diagnostic engine for intelligent identification of arrhythmias and respiratory abnormalities. Supports remote reporting and multi‑terminal connectivity.
5. ODM/OEM Manufacturing Capabilities
- Full‑cycle customization: product definition, industrial design, hardware/software development, mold opening.
- ISO 13485‑certified production base, flexible from pilot runs to mass production.
- FDA & CE applications and clinical data submitted. Registration certificates expected in June 2026.
Why Visit Booth 6.1D07?
- Hands‑on experience with our full product lineup
- Direct discussions with technical experts and international business team
- First look at new product iterations
- Dedicated international business zone for seamless cooperation inquiries
Booth Information
Dates: April 9–12, 2026
Venue: National Exhibition and Convention Center, Shanghai
Xinyan Medical Booth: 6.1D07
Join us in Shanghai to shape the future of cardiac care!
At CMEF Spring 2026, we look forward to welcoming you at Booth 6.1D07. Whether you are a clinician seeking advanced monitoring or a global partner exploring ODM/OEM opportunities, our team is ready to discuss how Xinyan’s innovations can support your mission.
Follow Xinyan Medical for more updates.
Follow us on WeChat